Clinical Research on Knee Osteoarthritis using MicoCell 3D Cell Culture Vessel -Patients were Injected with Cell Aggregates at our Collaborative Research Clinic-
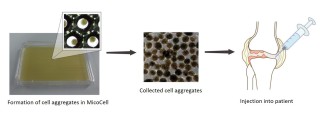
NIPPON SHOKUBAI CO., LTD. (Headquarters: Osaka, Japan, President: Yujiro Goto, hereinafter “Nippon Shokubai”) and Saiseikai Sobajima Clinic (Location: Osaka, Japan, Director: Satoshi Sobajima, hereinafter “Sobajima Clinic”) have been engaged in clinical research regarding treatment of knee osteoarthritis by injection of adipose-derived stem cell aggregates produced with the MicoCell; three-dimensional cell culture vessel developed by Nippon Shokubai since February 2021. Using this system, adipose-derived stem cell aggregates have now been administered to two patients at Sobajima Clinic.
Utilizing our unique technology, Nippon Shokubai developed MicoCell as a three-dimensional cell culture vessel that is capable of producing large quantities of cell aggregates with properties similar to those in the living body and a uniform particle size. Nippon Shokubai and Sobajima Clinic have been engaged in joint clinical research to obtain data on the safety and efficacy of human adipose-derived stem cell aggregates produced using MicoCell for treatment of knee osteoarthritis. In this clinical research, cell aggregates produced from the patient’s own adipose-derived stem cells using MicoCell are be injected into the knee joint. Sobajima Clinic submitted a Type 2 Regenerative Medicine Plan required by the Act on the Safety of Regenerative Medicine to the Regenerative Medicine Committee, which was approved on June 10, 2020, and accepted by the Kinki Regional Bureau of Health and Welfare on December 18, 2020. More details of this research are available in the Clinical Research Database.
Japan Registry of Clinical Trials
URL: https://jrct.niph.go.jp/
Clinical Research Protocol Number: jRCTb050200097.
In this clinical research at the Sobajima Clinic, we plan to confirm the safety of administering these cell aggregates produced using MicoCell in a total of five patients by March 2022. This clinical research will make significant contributions to the practical application of cell aggregation-based treatment and the further development of regenerative medicine.
Knee osteoarthritis (OA) involves the wear or loss of cartilage in the knee, leading to deformation of the knee, pain, and swelling. There are approximately 10 million patients in Japan, and over 33 million potential patients with no subjective symptoms in whom knee OA could be confirmed by X-ray diagnosis. Aging is the major cause of knee OA, and the number of patients is therefore increasing with the aging of the population.
Reference: “Report on Measures to Prevent Musculoskeletal Diseases to Promote Long-Term Care Prevention,” July 1, 2008, Study Group on Measures to Prevent Musculoskeletal Diseases to Promote Long-Term Care Prevention, Health and Welfare Bureau for the Elderly, Ministry of Health, Labour and Welfare.
MicoCell is the trademark of Nippon Shokubai.
About NIPPON SHOKUBAI Co., Ltd.: Since 1941, Nippon Shokubai has grown up its business with unique catalyst technology. Nippon Shokubai has supplied, for example, ethylene oxide, acrylic acid, automobile catalysts, process catalysts and so on. Among all, our global market share of superabsorbent polymers is the largest in the world now. Nippon Shokubai is a global chemical company operating under its corporate mission “TechnoAmenity – Providing affluence and comfort to people and society, with our unique technology.”
https://www.shokubai.co.jp/en/
【Contacts】
Corporate Communications Dept.,
NIPPON SHOKUBAI CO., LTD.
TEL: +81-3-3506-7605 E-mail: shokubai@n.shokubai.co.jp
[Files]