Biodegradable Chelating Agent: HIDS™ Tetrasodium 3-Hydroxy-2,2'-Iminodisuccinate
- Main Applications
Household detergents, Industrial detergents, Fiber surface conditioner, Bleaching aids, Water treatment agents, Chemical agents for photography, Paper and pulp processing additives, Plated sheet steel detergents, Semiconductor detergents, Anti-deposition for aluminum cases (aluminum etching), Plating pretreatment agents (degreasing agents, derusting agents), Soil improving agents, Raw materials for cosmetics, etc.
- Applications
- Functions
Basic Information
| Chemical Structure | 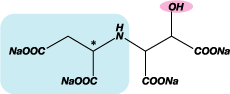 |
|---|---|
| Chemical Formula | C8H7NO9・4Na |
| CAS No. | 190195-65-4 |
| Molecular Weight | 353.1 |
| Appearance | Pale yellow~yellow, liquid |
| Product Form | 50wt% aqueous solution |
| pH | 10±1(*) |
| Density | 1.40~1.60 (20 C) |
Features and Properties
- High biodegradable chelating capability: HIDS™ effectively traps and inactivates heavy metal ions such as Fe3+ and Cu2+, as well as Ca2+ and Mg2+ over wide range of pH; it prevents troubles caused by metal ions contained in water.
- High performance under various conditions: HIDS™ shows superior performance as a chelating agent added to industrial and household detergents, etc. It has high thermal stability, high solubility in concentrated alkaline aqueous solutions, and low corrosion to glass.
- Environmentally-friendly chelating agent: HIDS™ is a top-class biodegradable and safe chelating agent.
Examples of Applications
HIDS™ has a wide-range of applications including in: Household detergents, Industrial detergents, Fiber surface conditioner, Bleaching aids, Water treatment agents, Chemical agents for photography, Paper and pulp processing additives, Plated sheet steel detergents, Semiconductor detergents, Anti-deposition for aluminum cases (aluminum etching), Plating pretreatment agents (degreasing agents, derusting agents), Soil improving agents, Raw materials for cosmetics, etc.
Please use this product after investigating by yourself matters concerning the compliance of legal regulations, independent industry standards and other rules and the proper usage methods for your intended applications.
Details of Functions
Acid Dissociation Constants and Stability Constants of Chelate Complexes
| Acid Dissociation Constants | pKa1:2.83 pKa2:3.56 pKa3:5.15 pKa4:9.11 |
|---|---|
| Stability Constants of Chelate Complexes | pKaCa2+:4.8 pKaFe3+:12.5 |
| C.V. Values | 300mg CaCO3/g |
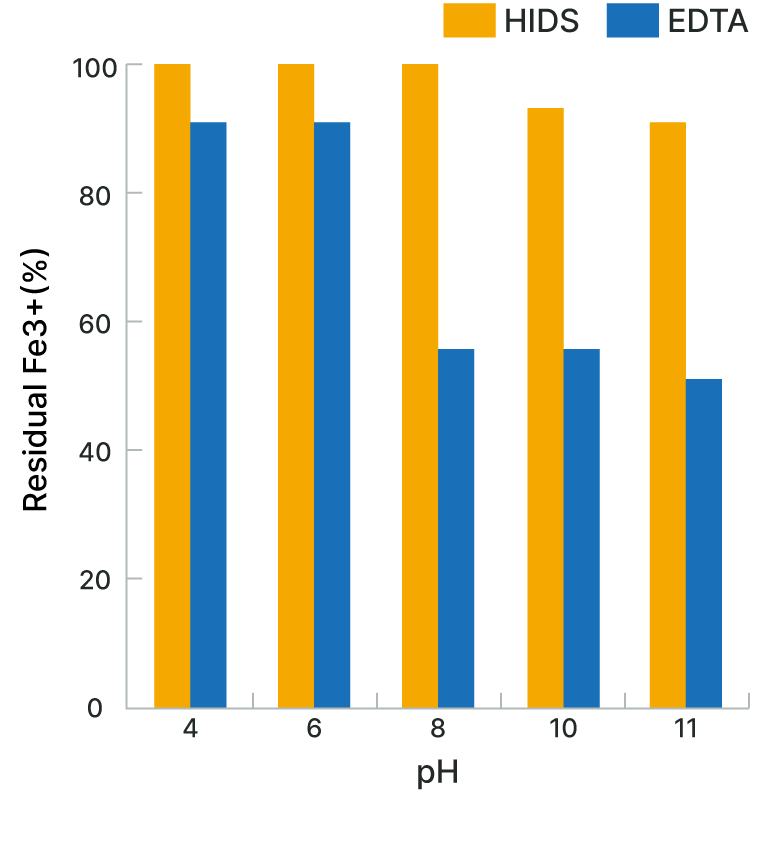
Fe3+ Chelating Capability
The biodegradable chelating agent HIDS™ forms stable water-soluble complexes with various metal ions over a wide range of pH values.
It is particularly excellent in chelating Fe3+ in alkaline solutions.
| Test Solution | Chelating agent 0.01M Fe3+ 0.01M |
|---|---|
| Temperature | Room temperature |
| Test Method | The test solution was left to stand for seven days at room temperature. Then the solution was filtered to remove the sediments (hydrate Fe compounds), and the Fe3+ concentration in the filtrate was measured. |
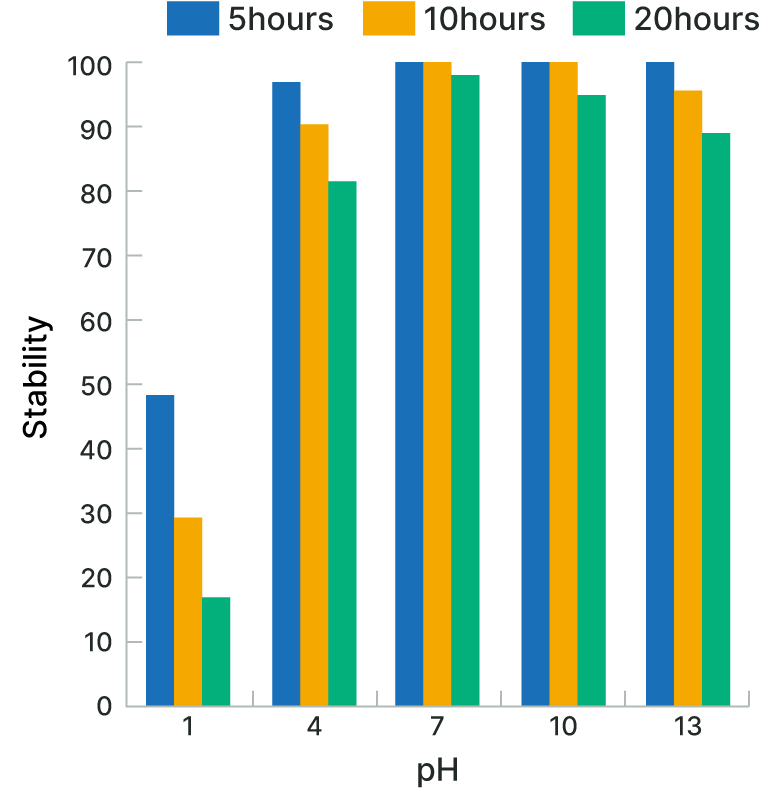
Thermal Stability and pH Stability
The biodegradable chelating agent HIDS™ has a wide range of applications including in harsh conditions; it is stable at high temperatures and at a range of pH 4-13.
| Test Solution | Biodegradable chelating agent HIDS™: 10% |
|---|---|
| Temperature | 80℃ |
| Test Method | The test solution was incubated at 80°C, and aliquots were sampled at the indicated hours. The residual amount of the biodegradable chelating agent HIDS™ was measured using high-performance liquid chromatography (HPLC). |
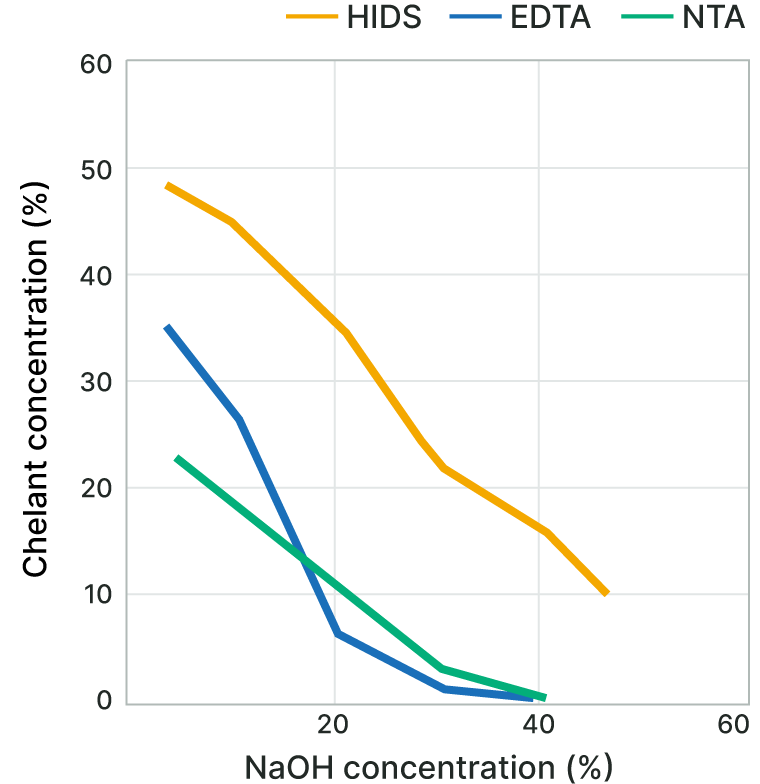
Solubility in Alkaline Aqueous Solutions
The biodegradable chelating agent HIDS™ is highly soluble in alkaline aqueous solutions.
In particular, HIDS™ can be added into highly-concentrated alkaline aqueous solutions without the addition of solubilizing agents.
| Temperature | 0℃ |
|---|
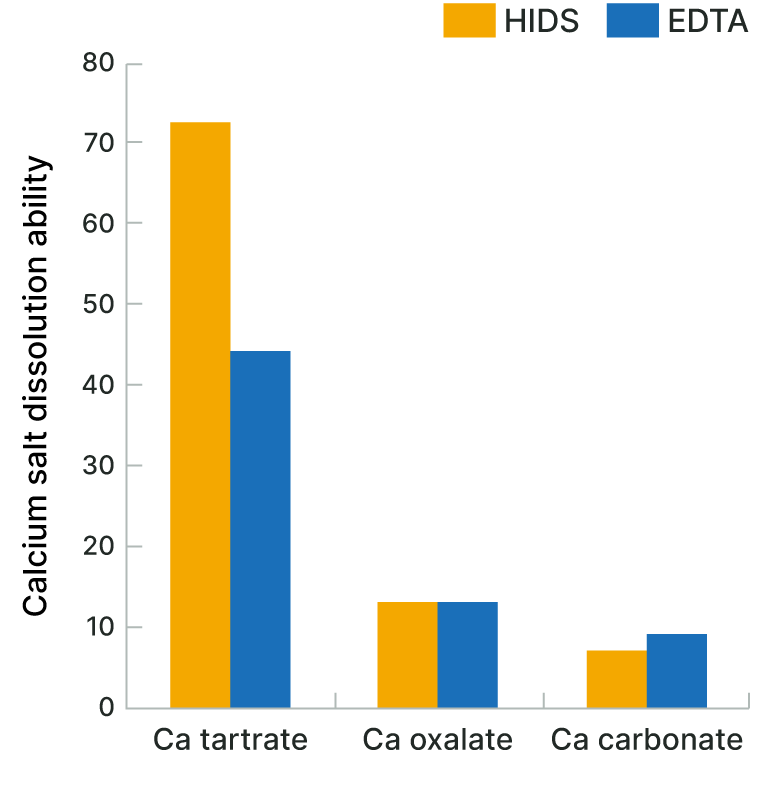
Solubilization of Water-insoluble Calcium Salt
The biodegradable chelating agent HIDS™ is able to solubilize water-insoluble calcium salt, which is the main component of scales.
HIDS™ can effectively remove scales in tanks and tubes, and prevent scale generation.
| Test Solution | Water-insoluble calcium salt: 0.6%, Chelating agent: 0.2%, NaOH: 3% |
|---|---|
| Temperature | 80℃ |
| Test Method | The test solution was stirred at 80°C for one hour, and then the concentration of dissolved Ca2+ was measured. |
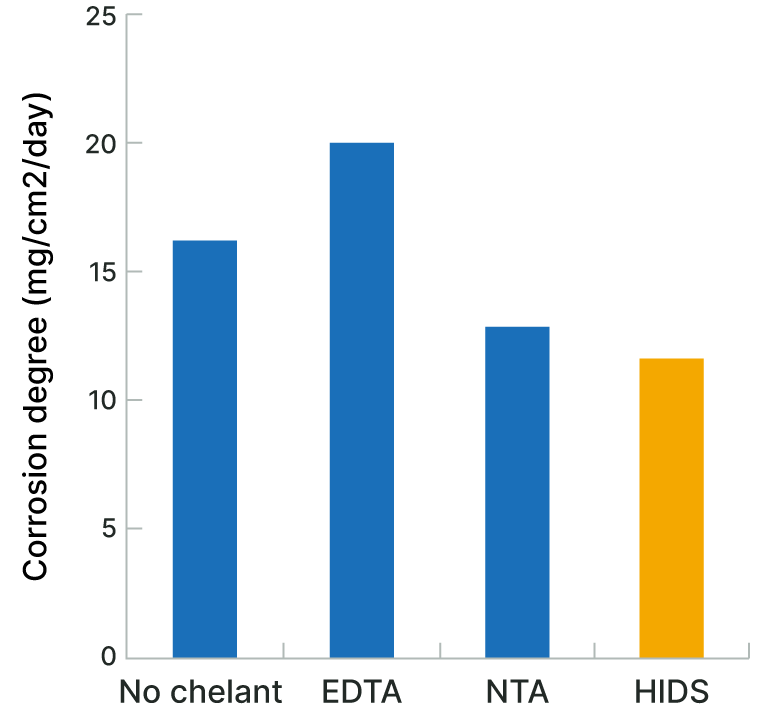
Glass Corrosion
The biodegradable chelating agent HIDS™ is less corrosive to glass compared to other chelating agents.
| Test Solution | NaOH 10%, Chelating agent 3% |
|---|---|
| Test Piece | Slide glass |
| Temperature | 100℃ |
| Test Method | The test pieces were placed in the test solution for three days at 100°C, and then removed from the solution and washed. The corrosion degree was calculated from the weight change per unit of area. |
Environmental Impact
| Biodegradability | Biodegradable (*1) |
|---|---|
| Fish Toxicity | LC50 ≥ 2,000 ppm (Himedaka (Oryzias latipes) 96 hr) (*1) |
Safety
| Acute Toxicity | Oral Toxicity LD50 > 2,000 mg/kg (*1) |
|---|---|
| Local Effect | No primary skin irritation (P.I.I. = 0.4 (45 wt%), P.I.I. = 0 (35 wt%), P.I.I. = 0 (10 wt%) P.I.I. = 0 (89 wt%, powder)) (*1) No primary eye irritation (*1) |
| Sensitization | No skin sensitization (*1) |
| Ames Test | Negative (*1) |
Laws and Regulations
| Chemical Substances Control Act (CSCA) | (2)-3803 |
|---|---|
| Industrial Safety and Health Act | 2-(4)-945 |
| TSCA | Listed in TSCA Inventory |
Handling and Storage Precautions
- Please use the products while wearing appropriate protective equipment, and avoid contact with the eyes, skin and clothing.
- Please avoid storing the products in direct sunlight, and keep the lids tightly closed for storage.
Protective Equipment
Eye protection: Safety glasses with side shields, goggles, plastic face shield
Hand protection: Protective (chemical resistant) gloves
Skin and Body protection: Safety clothes and shoes
The data listed above are extracted from the Material Safety Data Sheet (SDS, HPJ-1521-00-01).Safety information for this product is listed in the Chemical Products Safety Data Sheet (SDS: HPJ-1521-00-00); it is available on request from Nippon Shokubai.