In development Intermediate Water Polymer
- Main Applications
Biocompatible polymers
- Applications
Basic Information
Intermediate Water Polymer is a hydrophilic, biocompatible polymer developed by Nippon Shokubai through joint research with Professor Masaru Tanaka of the Institute for Materials Chemistry and Engineering (IMEC), Kyushu University.
What is Intermediate Water Polymer?
Professor Masaru Tanaka’s research revealed that polymer materials with excellent biocompatibility share a common characteristic : they contain intermediate water.*
Intermediate water polymers are polymer materials designed and developed to contain a large amount of intermediate water.

* Intermediate Water
When polymer materials are hydrated with water, the hydrated water takes on various states through its interaction with the polymers. These states are roughly grouped into three types depending on the degree of interaction: free water, intermediate water, and non-freezing water. Intermediate water is the middle state between free water and non-freezing water, and is water that gently interacts with polymers.
| Interaction with Polymers | Freezing Point | ||
|---|---|---|---|
| Free Water | Weak | 0℃ | Freezes and melts at 0°C, just like ordinary water. |
| Intermediate Water | Moderate | < 0°C | Gently interacts with polymer materials, and freezes and melts at temperatures under 0°C. |
| Non-freezing Water | Strong | Not detected | Does not freeze even at (minus) -150°C. |
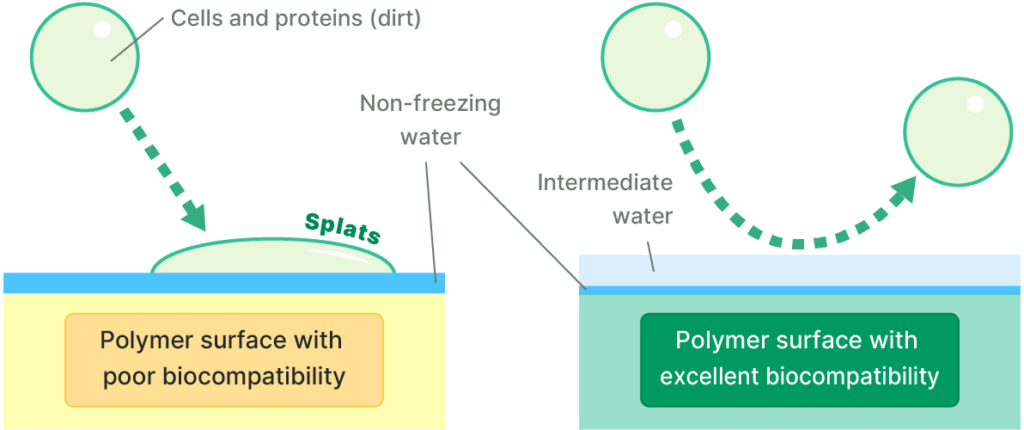
Mechanism of Biocompatibility
Biological foreign-body and rejection responses begin when cells recognize a material as foreign. Cells perceive their external environment primarily through proteins.
Because intermediate water is loosely structured, it remains on the polymer surface for an extended period and is believed to function as a cushion layer that inhibits direct contact between proteins or cells and the polymer surface. As a result, polymers rich in intermediate water are considered less likely to be recognized as foreign by the body.
On the other hand, reports (*) indicate that when proteins come into contact with non-freezing water formed on polymer surfaces, the proteins tend to undergo dehydration and denaturation. From the perspective of biocompatibility, non-freezing water is therefore considered undesirable.
Nippon Shokubai’s Intermediate Water Polymer is characterized by a high content of intermediate water and a low amount of non-freezing water.
* Journal of Biomaterials Science, Polymer Edition, Vol.3, p.127 (1992)